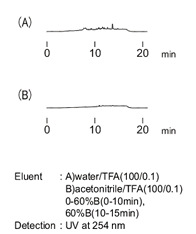
There was a case where during the blank run as a part of a preliminary study of gradient elution without a sample, an infinite number of peaks as in figure (A) appeared. When conducting a similar operation where the column was disconnected, the ghost peak disappeared as shown in figure (B). Consequently, the cause must be the column.
However, despite flushing or replacing the column, the baseline would not improve. Therefore many factors other than columns were examined; the cause was found to be water used to prepare eluents. Standard distilled water (inadequate for HPLC) had been mistakenly used, rather than HPLC grade distilled water which leads to an excellent baseline as in figure (B).
As discussed above, the water purity can have a great impact on gradient elution. Even HPLC grade distilled water will become contaminated as time passes after purchase, causing ghost peaks. A condition which has no significant influence on isocratic elution could cause a problem in gradient elution. In gradient elution, a column is equilibrated with an eluent with low content of organic solvent so that impurities in the eluent are adsorbed and concentrated in the column. After initiating analysis, content of organic solvent increases and impurities begin to be eluted resulting in the ghost peak. The heights of ghost peaks are dependent on the duration of equilibration. The ghost peak did not appear without a column because of the absence of adsorption and concentration during equilibration.
In gradient analysis, solvent requires great care especially of its grade distinction and storage.